3D printing and bioprinting strategies offer researchers the ability to engineer complex tissue constructs composed of multiple cellular, extracellular matrix, and biomolecular components in an effort to recapitulate the complexity of native tissue physiology. These 3D printed tissues can be applied to better understand the mechanistic underpinnings of the in vivo tissue environment in a controlled in vitro platform, develop disease models and their treatment using promising drug candidates, and even fabricate implantable tissues for patients in need. However, another critical aspect of native physiology is its inherent dynamic nature, such as pressurized blood flow through the vasculature, the stretching and relaxation of muscle fibers, and the tension and compression subjected to our bones and joints during every physical movement. In order for the 3D printed constructs to reflect native conditions, it is imperative that these dynamics are also recreated in our in vitro platforms.
To address this concern, TR&D1 focuses on developing 3D printed bioreactors as a dynamic culture system to control the cellular microenvironment that promotes cell bioactivity within large, engineered constructs. Specialized bioreactor chambers are fabricated using 3D printing techniques to engineer variable architecture, controlled flow environments, and spatially located cell populations that enable adequate availability of nutrients, and enable cell-cell interactions, within these large constructs. The bioreactors can be also engineered to subject 3D printed constructs to physical tension or compression as dictated by tissue needs. The versatility of 3D printing allows us to engineer these bioreactors for both 2D and 3D culture across a broad range of biomaterials, cell types, and tissue applications.
This TR&D provides a strong foundation and collaborative support to fabricate removable, biodegradable scaffolds of engineered tissues that are suitable for in vivo applications and to facilitate the development of other similarly designed, tissue-specific bioreactor systems.
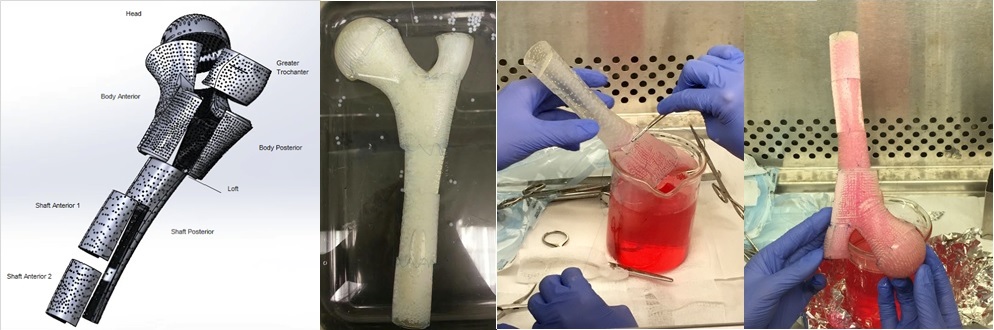
[Dynamic Bioreactor Culture of High Volume Engineered Bone Tissue. Nguyen et al., Tissue Engineering Part A. 22: 263-271 (2016).]
Related Links
Images courtesy of UMD.